Can Cord Blood Cure Autism?
When your child was born, you may have counted his fingers and toes and checked him for other signs of health. But not all medical conditions are immediately obvious judging by physical appearance alone. If your child struggles with social interactions and communication, it is possible that he has autism spectrum disorder (ASD), a developmental disorder. Researchers have long sought to cure autism, but to date, there is no magic bullet. Now, the Sutter Neuroscience Institute in Sacramento, CA, in collaboration with the Cord Blood Registry, has announced an FDA-regulated clinical trial to evaluate a new way to treat or cure autism.
The study has been planned by Dr. Michael Chez and his colleagues for over a year. It is unique because it is the first clinical trial regulated by the FDA to study the use of cord blood stem cells to treat or cure autism. In particular, the researchers plan to evaluate what effect, if any, the treatment has upon the study participants’ speech and language, as well as their behaviors.
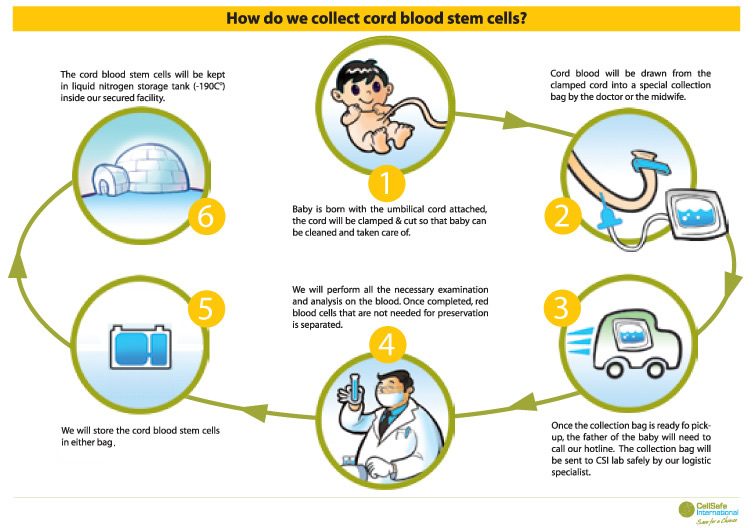
Dr. Chez and his colleagues have carefully screened the study participants. They have selected 30 children with autism, all of whom have been screened for other medical conditions that might influence the results, including head injury, premature birth, and Fragile X syndrome. All of the children have had their cord blood stored by the Cord Blood Registry. Dr. Chez has also removed another significant factor from the study that may have influenced the results: genetic predisposition to autism. None of the children have genetic markers for autism, which means that they presumably developed it because of other factors.
This study has been deemed to be safe and ethical because the children will receive infusions of their own cord blood. This eliminates the possibility that their bodies will reject the infusions. The children will be divided into two groups. The first group will receive one infusion of cord blood at the beginning of the trial. The second group will receive a placebo saline infusion. Neither the children nor the researchers administering the infusions will know which children will receive which infusions.
The kids will be monitored for six months, however Dr. Chez has expressed optimism that some results will be evident within six weeks. At the six-month mark, the groups will reverse their roles. Group one will receive a saline infusion, and group two will receive a cord blood infusion. The groups will again be monitored.
Dr. Chez has theorized that cord blood could potentially help children with autism because some children with ASD have dysfunctional immune systems. This can potentially damage the nervous system or affect its development. The cord blood stem cells are intended to repair the immune system, which may theoretically improve symptoms of autism like speech and language impairments.
For more information on the clinical trial, check out the press release from the Cord Blood Registry. If you believe your child might have a speech disorder or delay that might be caused by autism or another condition, have him evaluated by a certified speech-language pathologist (SLP). There are many speech therapy tools that can help your child, from Speech Buddies to voice therapy.